A Unified Route to the Welwitindolinone Alkaloids: Total Syntheses of (−)-N-Methylwelwitindolinone C Isothiocyanate, (−)-N-Methylwelwitindolinone C Isonitrile, and (−)-3-Hydroxy-N-methylwelwitindolinone C Isothiocyanate
Kevin M. Allan, Kenichi Kobayashi, and Viresh H. Rawal
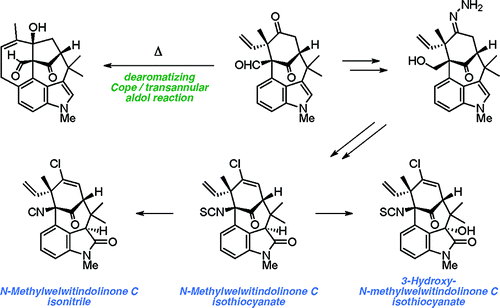
As part of a comprehensive strategy to the welwitindolinone alkaloids possessing a bicyclo[4.3.1]decane core, we report herein concise asymmetric total syntheses of (−)-N-methylwelwitindolinone C isothiocyanate (2a), (−)-N-methylwelwitindolinone C isonitrile (2b), and (−)-3-hydroxy-N-methylwelwitindolinone C isothiocyanate (3a) from a common tetracyclic intermediate. The crucial vinyl chloride moiety was installed through electrophilic chlorination of a hydrazone, but only after adjustment of reactivity to circumvent a facile skeletal rearrangement. Selective desulfurization and oxidation of 2a provided access to 2b and 3a, respectively. Notably, this work provides corrected 1H and 13C NMR spectral data for 3a.
JOURNAL OF THE AMERICAN CHEMICAL SOCIETY 134: 3 (28 Dec. 2011): 1392–95